Delivering person centred care in long-term conditions
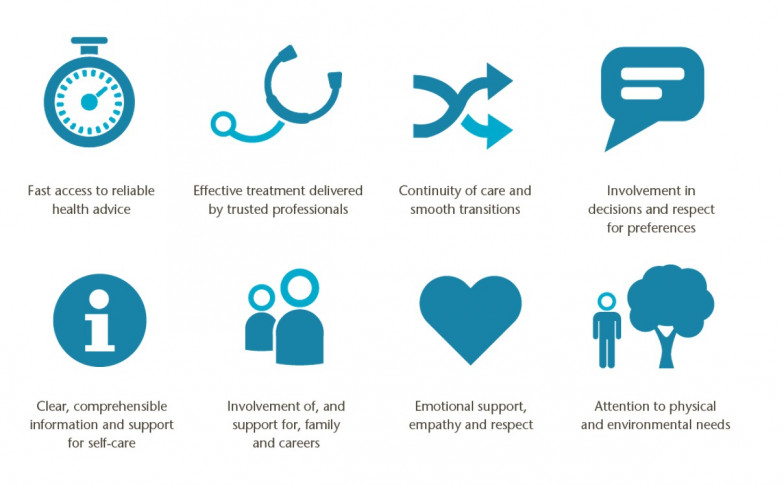
Transforming support for people with long term conditions makes sense from every angle: improving wellbeing, developing a more positive sense of control, achieving better health outcomes, and potentially lower healthcare costs. It now seems possible to envisage this as the norm but substantial barriers and challenges exist. We need clinical and patient leadership to help navigate these challenges, along with system drivers and incentives, and practical support to encourage these new ways of working. People with lower activation levels, poor health literacy, and difficult social circumstances need specific and tailored support but have the most to gain.11 We believe that the success of any clinician, team, or organisation should be defined by their efforts to tackle these disadvantages and to ensure a person’s goals and priorities are always at the centre of his or her care.
Read more about the article here.(external link)
Patient commentary: What I need to self manage my care
Alex, a 27 year old with type 1 since age 18 months talks about what's helped her and what's needed for patient-centred care.
What helps:
- Medical devices and repeat prescriptions
- Multidisciplinary healthcare team in secondary care
- Education
- Empathy
- Engagement
- Integrated services
"For me the next logical steps are to create my own care plan, containing goals set and measured by me and accessible at a time and location that suits me (online or offline), and to work with the stakeholders I choose, from the list mentioned above, so that I can achieve and update my care plan."
What doesn't help:
"What I don’t want is to be given routine outpatient appointments. People don’t want to spend time in clinics to manage their health; they want to do it at home in a way that suits them, like their banking or shopping. This is person-centered care and in some places it’s already happening."
What's needed:
But we will only get universal person-centered care when we begin valuing, measuring, and responding to the things that patients really care about – and doing so in a systematic, fully integrated 'whole system' way." (1)
Read the full BMJ article(external link).
Patients as partners – a guide
Building collaborative relationships among professionals, patients, carers and communities.
Key messages
The guide suggests five approaches to developing an effective relationship:
-
- find your collaborative partner: this, together with identifying funding and a real task to work on, marks the first important stage of beginning a collaborative relationship
- invest in developing leadership and collaborative relationships
- make time for learning: set aside time for reflection, record learning and share that learning with peers and those you hope to influence
- go where the energy is: sometimes this means working under the radar for a while until you are ready to share your results, your approach and your learning
- embed collaborative activity at all levels in your system or organisation.
Source: The Kings Fund – ideas that change healthcare, UK
Patient–centered care: the key to cultural competence
This article summarises the key principles of patient–centered cultural competence and relevant skills, phrases and examples.
Read full article here(external link).
Source: Annals of Oncology, Oxford Academic.
Ten commandments for patient-centred treatment
Lehman, R. Tejani, A. et al. Ten Commandments for patient-centred treatment.(external link) Br J Gen Pract Oct 2015, 65 (639) 532-533
References
- Silverstein A. Patient commentary: What I need to self-manage my care(external link) BMJ 2015; 350 :h248
- Lehman, R. Tejani, A. et al. Ten Commandments for patient-centred treatment.(external link) Br J Gen Pract Oct 2015, 65 (639) 532-533